Nanotheranostics 2021; 5(1):57-72. doi:10.7150/ntno.50095 This issue Cite
Research Paper
Intraoperative assessment and postsurgical treatment of prostate cancer tumors using tumor-targeted nanoprobes
1. Department of Neurosurgery, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA.
2. Biomedical Imaging Research Institute, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA.
3. Department of Medicine, Uro-Oncology Research Program, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA.
4. S. Mark Taper Foundation Imaging Center, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA.
5. Samuel Oschin Comprehensive Cancer Institute, Cedars-Sinai Medical Center, Los Angeles, CA 90048, USA.
6. Current address: Department of Cell Biology and Biochemistry, Texas Tech University Health Sciences Center, Lubbock, TX 79430, USA.
Abstract
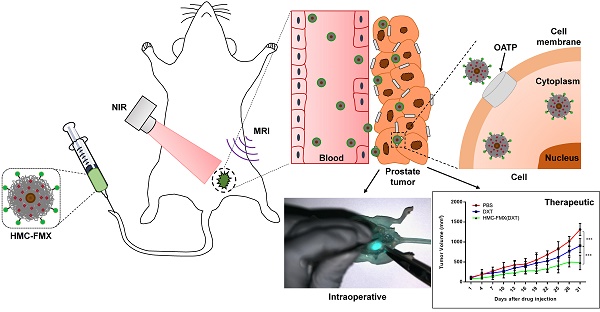
Successful visualization of prostate cancer (PCa) tumor margins during surgery remains a major challenge. The visualization of these tumors during surgery via near infrared fluorescence (NIRF) imaging would greatly enhance surgical resection, minimizing tumor recurrence and improving outcome. Furthermore, chemotherapy is typically administered to patients after surgery to treat any missed tumor tissue around the surgical area, minimizing metastasis and increasing patient survival. For these reasons, a theranostics fluorescent nanoparticle could be developed to assist in the visualization of PCa tumor margins, while also delivering chemotherapeutic drug after surgery.
Methods: Ferumoxytol (FMX) conjugated to the fluorescent dye and PCa targeting agent, heptamethine carbocyanine (HMC), yielded the HMC-FMX nanoprobe that was tested in vitro with various PCa cell lines and in vivo with both subcutaneous and orthotopic PCa mouse models. Visualization of these tumors via NIRF imaging after administration of HMC-FMX was performed. In addition, delivery of chemotherapeutic drug and their effect on tumor growth was also assessed.
Results: HMC-FMX internalized into PCa cells, labeling these cells and PCa tumors in mice with near infrared fluorescence, facilitating tumor margin visualization. HMC-FMX was also able to deliver drugs to these tumors, reducing cell migration and slowing down tumor growth.
Conclusion: HMC-FMX specifically targeted PCa tumors in mice allowing for the visualization of tumor margins by NIRF imaging. Furthermore, delivery of anticancer drugs by HMC-FMX effectively reduced prostate tumor growth and reduced cell migration in vitro. Thus, HMC-FMX can potentially translate into the clinic as a nanotheranostics agent for the intraoperative visualization of PCa tumor margins, and post-operative treatment of tumors with HMC-FMX loaded with anticancer drugs.
Keywords: image-guided therapy, near infrared fluorescence imaging, iron oxide nanoparticles, heptamethine cyanine, prostate cancer

 Global reach, higher impact
Global reach, higher impact