Nanotheranostics 2017; 1(2):154-165. doi:10.7150/ntno.19449 This issue Cite
Research Paper
Nanoliposomes for Safe and Efficient Therapeutic mRNA Delivery: A Step Toward Nanotheranostics in Inflammatory and Cardiovascular Diseases as well as Cancer
1. Department of Thoracic, Cardiac and Vascular Surgery, Clinical Research Laboratory, University Hospital Tübingen, Germany;
2. Atherothrombosis and Vascular Biology, Baker Heart & Diabetes Institute, Melbourne, Victoria, Australia;
3. Department of Medicine, Monash University, Melbourne, Victoria, Australia.
* Equally contributing last authors
Abstract
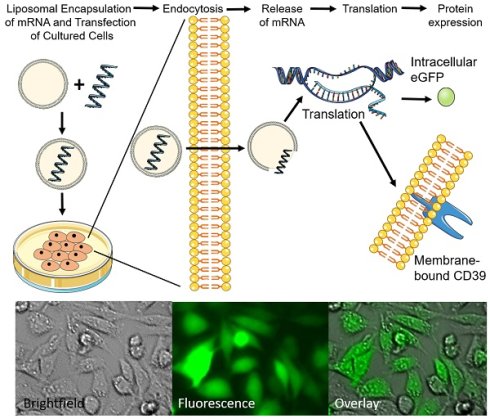
Rationale: Genetic therapy using modified mRNA for specific therapeutic protein expression for disease treatment and vaccination represents a new field of therapeutic and diagnostic medicine. Non-viral vectors transfection using biocompatible nanoliposomes enables safe and efficient delivery of therapeutic mRNA.
Objective: Generation of non-toxic, cell-compatible cationic nanoliposomes as nanotheranostic agents to successfully deliver therapeutic mRNA.
Methods and results: Cationic nanoliposomes (DC-Cholesterol/DOPE) were generated as transfection vehicles for either eGFP mRNA or the therapeutic anti-inflammatory, CD39 mRNA. We observed no toxicity using these nanoplexes and noted high cell viability after transfection. Nanoplexes for the transfection of eGFP mRNA showed an increase in fluorescence signals on microscopy as compared to the mRNA control after 24 hours in Chinese hamster ovary (CHO) cells (14.29 ± 5.30 vs. 1.49 ± 0.54; mean ± SD respectively; p<0.001) and flow cytometry (57.29 ± 14.59 vs 1.83 ± 0.34; % mean ± SD; p<0.001). Nanoplexes for the transfection of CD39 mRNA showed increased CD39 expression in flow cytometry (45.64 ± 15.3 vs. 3.94 ± 0.45; % mean ± SD; p<0.001) as compared to the mRNA control after 24 hours using CHO cells. We also demonstrated efficient transfection across several cell lines (CHO, HEK293, and A549), as well as long-term protein expression (120 h and 168 h) using these nanoplexes.
Conclusions: We have developed and tested non-toxic, safe, and efficient nanoliposome preparations for the delivery of therapeutic mRNA that hold promise for novel therapies in diseases such as inflammatory and cardiovascular diseases, as well as cancer. We have also demonstrated that this approach provides a reliable technology to deliver CD39 mRNA as an anti-inflammatory therapeutic for future nanotheranostics approaches.
Keywords: anti-inflammatory, mRNA, nanoliposomes, therapeutics, transfection.

 Global reach, higher impact
Global reach, higher impact