Nanotheranostics 2023; 7(4):412-423. doi:10.7150/ntno.84574 This issue Cite
Research Paper
Synthesis of polycationic nanoparticles for microbial inhibition and killing
1. Polymeric Materials Area, Chemical and Material Sciences Division, CSIR-Indian Institute of Petroleum, Dehradun-248005, India.
2. Academy of Scientific and Innovative Research (AcSIR), Ghaziabad-201002, India.
3. Biochemical Science Area, Material Resource Efficiency Division, CSIR-Indian Institute of Petroleum, Dehradun-248005, India.
Abstract
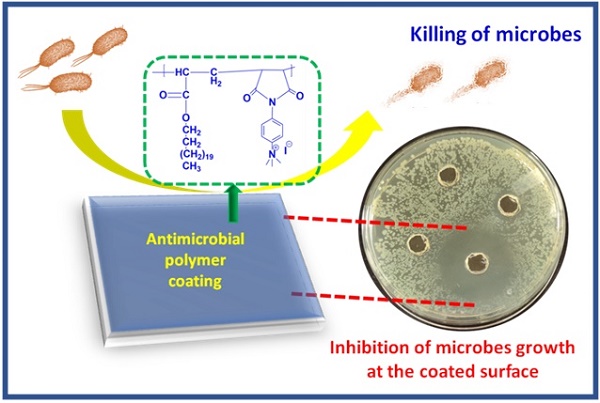
Antimicrobial polymers (AMP) appear to be a promising candidate to deal with the current scenario of bacterial resistance against conventional drugs and antibiotics as they mainly depend on disrupting the bacterial membrane. This work investigates the effect of polycations bearing aromatic and aliphatic pendant cationic groups on the antimicrobial performance of AMP. A radical polymerization strategy was adopted to synthesize two different copolymers and convert them into polycations upon post-modification. Polyelectrolytes were converted into nanoparticles by nanoprecipitation and named PE1 and PE2. Polymers were analyzed by NMR, FT-IR, and gel permeation chromatography (GPC). PE1 and PE2 nanoparticles were uniform, spherical particles from FESEM, size, and zeta potential measurements. The antimicrobial properties of polyelectrolytes were determined against pathogenic Escherichia coli (E. coli), Bacillus Subtilis (B. Subtilis), Bacillus Amyloliquefaciens (B. Amyloliquefaciens) and Citrobecter Freundii (C. Freundii) bacterias. The biocidal activity determination studies showed that polyelectrolyte PE2 with aromatic pendant units outperformed PE1 with the aliphatic pendant group. This work highlights the remarkable effect of aromatic segmentation, which provides microbial inhibition, and killing is demonstrated as an antibacterial surface coating.
Keywords: Polyelectrolytes, nanoparticles, biocidal activity, antimicrobial coatings

 Global reach, higher impact
Global reach, higher impact