Nanotheranostics 2022; 6(4):388-399. doi:10.7150/ntno.71906 This issue Cite
Research Paper
Covalent core-radiolabeling of polymeric micelles with 125I/211At for theranostic radiotherapy
1. Center for Nanomedicine and Theranostics (The Hevesy Laboratory), DTU Health Technology, Technical University of Denmark (DTU), Ørsteds Plads 345C, 2800 Lyngby, Denmark
2. Department of Drug Design and Pharmacology, Faculty of Health and Medical Sciences, University of Copenhagen, Universitetsparken 2, 2100 Copenhagen, Denmark
3. Departments of Radiation Physics, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gula Stråket 2b, 41345 Gothenburg, Sweden
4. Department of Clinical Physiology, Nuclear Medicine & PET, Rigshospitalet, Blegdamsvej 9, 2100 Copenhagen, Denmark
5. National Centre for Nano Fabrication and Characterization, Technical University of Denmark (DTU), Oersteds Plads—Building 347, 2800 Lyngby, Denmark
6. Cluster for Molecular Imaging, Department of Biomedical Sciences, University of Copenhagen, Blegdamsvej 3, 2100 Copenhagen, Denmark
Abstract
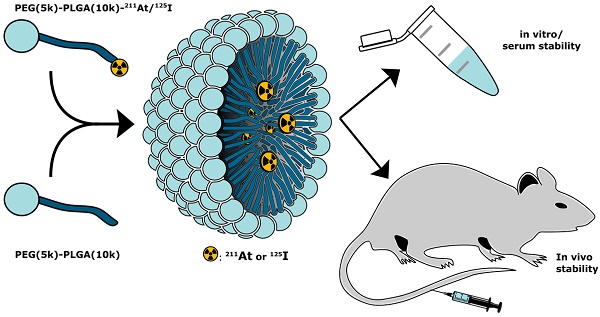
Astatine-211 (211At) is one of the most promising α-emitters for targeted alpha therapy, especially of cancer metastases. However, the lack of a stable isotope, frequent in vivo deastatination, and limited radiochemical knowledge makes it challenging to apply. Here, we report a new strategy for radiolabeling the lipophilic core of polymeric micelles (PMs) with covalently bound 211At. The PMs were radiolabeled via either an indirect synthon-based method or directly on the amphipathic block copolymer. The radiochemistry was optimized with iodine-125 (125I) and then adapted for 211At, enabling the use of both elements as a potential theranostic pair. PMs that were core-radiolabeled with both 125I or 211At were prepared and characterized, based on a PEG(5k)-PLGA(10k) co-polymer. The stability of the radiolabeled PMs was evaluated in mouse serum for 21 h, showing radiochemical stability above 85%. After in vivo evaluation of the 211At- labeled PMs, 4-5 % ID/g of the 211At could still be detected in the blood, showing a promising in vivo stability of the PMs. Further, 211At-labeled PMs accumulated in the spleen (20-30 %ID/g) and the liver (2.5- 5.5 %ID/g), along with some detection of 211At in the thyroid (3.5-9 %ID/g). This led to the hypothesis that deastatination takes place in the liver, whereas good stability of the 211At core-radiolabel was observed in the blood.
Keywords: Astatine-211, polymeric micelles, PEG-PLGA, alpha-therapy, iodine-125

 Global reach, higher impact
Global reach, higher impact