Nanotheranostics 2021; 5(1):1-7. doi:10.7150/ntno.47226 This issue Cite
Research Paper
Cancer-derived EVs show tropism for tissues at early stage of neoplastic transformation
1. Department of Health Sciences, Center of Excellence on Neurodegenerative Diseases, University of Milan, Italy.
2. Current address: Department of Pharmaceutical and Pharmacological Sciences, University of Padova, Italy.
3. Department of Pharmacological and Biomolecular Sciences, University of Milan, Milan, Italy.
4. Targovax Oy, Clinical Science, Helsinki, Finland.
5. National Institute of Public Health - National Institute of Hygiene, Department of Virology, Warsaw, Poland.
6. Department of Oncology and Hemato-Oncology, University of Milan, Milan, Italy.
7. Istituto Nazionale Tumori Fondazione IRCCS, National Cancer Institute, Milan, Italy.
#These authors contributed equally to this work.
Abstract
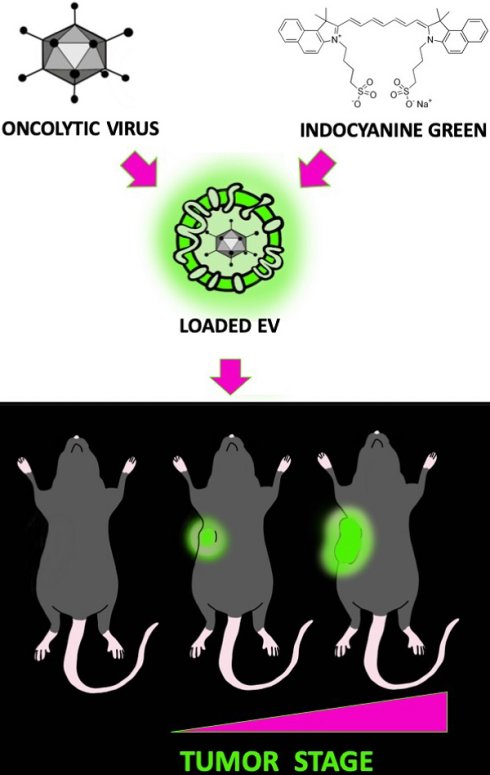
From the past decade, extracellular vesicles (EVs) have attracted considerable attention as tools for the selective delivery of anti-neoplastic drugs to cancer tissues. Compared to other nanoparticles, EVs display interesting unique features including immune compatibility, low toxicity and the ability to encapsulate a large variety of small- and macro-molecules. However, in virtually all studies, investigations on EVs have been focused on fully transformed cancers: the possibility to apply EV technology also to early-stage tumors has never been explored.
Methods: Herein, we studied the ability of cancer-derived EVs to recognize and deliver their cargo also to incipient cancers. To this purpose, EV biodistribution was studied in MMTV-NeuT genetically modified mice during early mammary transformation, in fully developed breast tumors and in the normal gland of wild type syngeneic mice. EVs were loaded with indocyanine green (ICG), a near-infrared (NIR) dye together with oncolytic viruses and i.v. injected in mice. The nanoparticle biodistribution was assayed by in vivo and ex vivo optical imaging (detecting the ICG) and semiquantitative real-time PCR (measuring the adenoviral genome) in different tissues.
Results: Our results demonstrate the ability of cancer-derived EVs to recognize early-stage neoplastic tissues opening the possibility to selectively deliver theranostics also for tumor prevention.
Conclusions: Taken together our study demonstrates the ability of EVs to recognize and deliver diagnostic and therapeutic agents not only to fully transformed tissues but also to early stage tumors. These findings pave the way for the synthesis of “universal” EVs-based formulation for targeted cancer therapy.
Keywords: Extracellular vesicles, oncolytic viruses, early-stage cancer therapy, drug delivery, in vivo imaging

 Global reach, higher impact
Global reach, higher impact