Nanotheranostics 2018; 2(4):387-402. doi:10.7150/ntno.26969 This issue Cite
Research Paper
A Scalable Method for Squalenoylation and Assembly of Multifunctional 64Cu-Labeled Squalenoylated Gemcitabine Nanoparticles
1. Department of Biomedical Engineering, University of California Davis, Davis, California, 95616, USA.
2. Department of Radiology, Stanford University, Palo Alto, CA 94304, USA
3. Department of Molecular and Cellular Biology, University of California Davis, Davis, California, 95616, USA.
Abstract
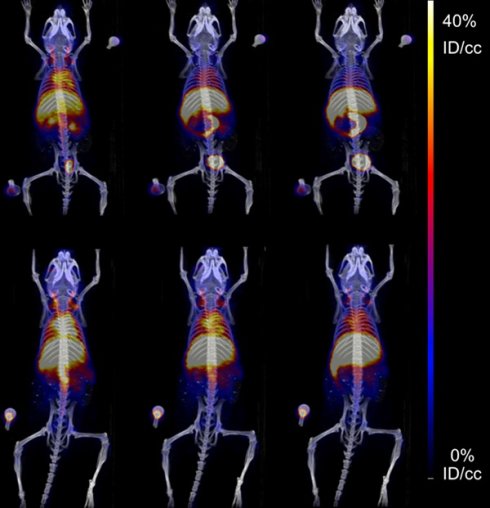
Squalenoylation of gemcitabine, a front-line therapy for pancreatic cancer, allows for improved cellular-level and system-wide drug delivery. The established methods to conjugate squalene to gemcitabine and to form nanoparticles (NPs) with the squalenoylated gemcitabine (SqGem) conjugate are cumbersome, time-consuming and can be difficult to reliably replicate. Further, the creation of multi-functional SqGem-based NP theranostics would facilitate characterization of in vivo pharmacokinetics and efficacy.
Methods: Squalenoylation conjugation chemistry was enhanced to improve reliability and scalability using tert-butyldimethylsilyl (TBDMS) protecting groups. We then optimized a scalable microfluidic mixing platform to produce SqGem-based NPs and evaluated the stability and morphology of select NP formulations using dynamic light scattering (DLS) and transmission electron microscopy (TEM). Cytotoxicity was evaluated in both PANC-1 and KPC (KrasLSL-G12D/+; Trp53LSL-R172H/+; Pdx-Cre) pancreatic cancer cell lines. A 64Cu chelator (2-S-(4-aminobenzyl)-1,4,7-triazacyclononane-1,4,7-triacetic acid, NOTA) was squalenoylated and used with positron emission tomography (PET) imaging to monitor the in vivo fate of SqGem-based NPs.
Results: Squalenoylation yields of gemcitabine increased from 15% to 63%. Cholesterol-PEG-2k inclusion was required to form SqGem-based NPs using our technique, and additional cholesterol inclusion increased particle stability at room temperature; after 1 week the PDI of SqGem NPs with cholesterol was ~ 0.2 while the PDI of SqGem NPs lacking cholesterol was ~ 0.5. Similar or superior cytotoxicity was achieved for SqGem-based NPs compared to gemcitabine or Abraxane® when evaluated at a concentration of 10 µM. Squalenoylation of NOTA enabled in vivo monitoring of SqGem-based NP pharmacokinetics and biodistribution.
Conclusion: We present a scalable technique for fabricating efficacious squalenoylated-gemcitabine nanoparticles and confirm their pharmacokinetic profile using a novel multifunctional 64Cu-SqNOTA-SqGem NP.
Keywords: Nanoparticles, gemcitabine, squalene, radiolabeling, 64Cu, pancreatic cancer

 Global reach, higher impact
Global reach, higher impact