Nanotheranostics 2022; 6(3):337-349. doi:10.7150/ntno.71510 This issue Cite
Research Paper
Surface enhanced deep Raman detection of cancer tumour through 71 mm of heterogeneous tissue
1. School of Physics and Astronomy, University of Exeter, Exeter EX4 4QL, UK
2. School of Health and Life Sciences, Teesside University, Middlesbrough TS1 3BX, UK
3. School of Pharmacy, University College London, London, UK
4. Central Laser Facility, Research Complex at Harwell, STFC Rutherford Appleton Laboratory, UK Research and Innovation, Harwell Campus OX11 0QX, UK
5. NanoPhotonics Centre, Cavendish Laboratory, Cambridge, CB3 0HE, UK
Abstract
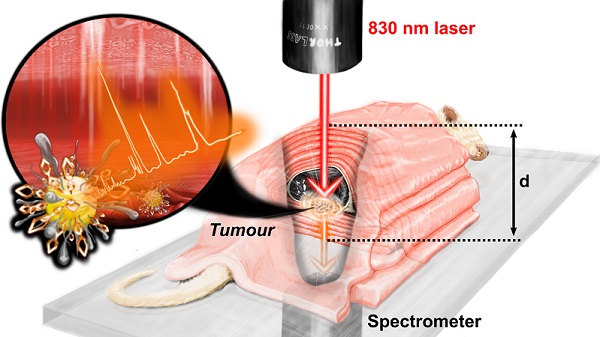
Detection of solid tumours through tissue- from depths relevant to humans- has been a significant challenge for biomedical Raman spectroscopy. The combined use of surface enhanced Raman scattering (SERS) imaging agents with deep Raman spectroscopy (DRS), i.e., surface enhanced deep Raman spectroscopy (SEDRS), offer prospects for overcoming such obstacles. In this study, we investigated the maximum detection depth through which the retrieval of SERS signal of a passively targeted biphenyl-4-thiol tagged gold nanoparticle (NP) imaging agent, injected subcutaneously into a mouse bearing breast cancer tumour, was possible. A compact 830 nm set-up with a hand-held probe and the flexibility of switching between offset, transmission and conventional Raman modalities was developed for this study. In vivo injection of the above SERS NP primary dose allowed surface tumour detection, whereas additional post mortem NP booster dose was required for detection of deeply seated tumours through heterogeneous animal tissue (comprising of proteins, fat, bone, organs, blood, and skin). The highest detection depth of 71 mm was probed using transmission, translating into a ~40% increase in detection depth compared to earlier reports. Such improvements in detection depth along with the inherent Raman chemical sensitivity brings SEDRS one step closer to future clinical cancer imaging technology.
Keywords: Plasmonic gold nanoparticles, SERS, SORS, TRS, SESORS, SEDRS mouse breast cancer, cancer tumour detection.

 Global reach, higher impact
Global reach, higher impact