Nanotheranostics 2022; 6(3):230-242. doi:10.7150/ntno.61280 This issue Cite
Research Paper
Mild hyperthermia accelerates doxorubicin clearance from tumour-extravasated temperature-sensitive liposomes
1. Nanomedicine Lab, UCL School of Pharmacy, University College London, Brunswick Square, London WC1N 1AX, United Kingdom.
2. School of Pharmacy, Queen's University Belfast, United Kingdom.
3. Faculty of Medical &Human Sciences and National Graphene Institute, University of Manchester, Manchester, United Kingdom.
Abstract
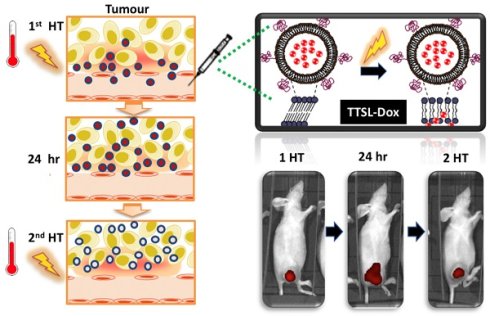
Mild hyperthermia (HT) (40-43 °C) has been combined with temperature-sensitive liposomes (TSL), offering on-demand drug release for increased drug bioavailability and reduced systemic toxicity. Different HT regimens have been applied to trigger liposome drug release in the blood vessels (intravascular) of heated tumours or following tumour extravasation (interstitial). The present study systematically assessed the in vivo doxorubicin (Dox) release and therapeutic efficacy of Dox-loaded TSL with different release profiles. Low temperature-sensitive liposomes (LTSL-Dox), traditional-temperature-sensitive liposomes (TTSL-Dox), and non-temperature-sensitive liposomes (NTSL-Dox) were combined with a single or two HT in different tumour models (murine melanoma B16F10 tumour and human breast MDA-MB-435). The efficacy of each treatment was assessed by monitor tumour growth and mice survival. The level of Dox in tumour tissues was quantified using 14C-Dox and liquid scintillation while Dox release was assessed using live imaging and confocal laser scanning microscopy. Applying a second HT to release Dox from extravasated TTSL-Dox was not therapeutically superior to single HT application due to Dox clearance from the extravasated TTSL-Dox. Our findings revealed that enhanced blood perfusion in heated tumours during the second water bath HT could be seen as a hurdle for TTSL-Dox's anticancer efficacy, where the systemic toxicity of the redistributed Dox from the tumour tissues could be potentiated.
Keywords: Intravascular release, interstitial release, hyperthermia, temperature-sensitive liposomes (TSL), doxorubicin, cancer

 Global reach, higher impact
Global reach, higher impact