Nanotheranostics 2022; 6(2):161-174. doi:10.7150/ntno.62639 This issue Cite
Research Paper
Development of CD44E/s dual-targeting DNA aptamer as nanoprobe to deliver treatment in hepatocellular carcinoma
1. Department of Surgery at Sir Y.K. Pao Center for Cancer, The Chinese University of Hong Kong, Shatin, Hong Kong, China.
2. Department of Biomedical Engineering, The Chinese University of Hong Kong, Shatin, Hong Kong, China.
3. Science Research Center, The Seventh Affiliated Hospital of Sun Yat-sen University, Shenzhen 518107, China.
4. State Key Laboratory of Translational Oncology, The Chinese University of Hong Kong, Shatin, Hong Kong, China.
Abstract
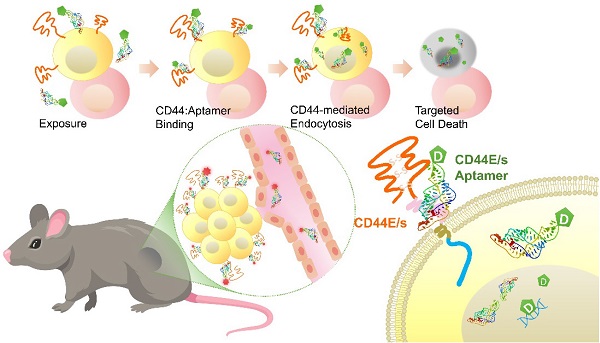
Background: Hepatocellular carcinoma (HCC) is the predominant subtype of liver cancer with an extraordinary high mortality. Resistance to systemic therapy is a major cause of inferior clinical outcome in most patients with HCC. CD44 is a transmembrane cell-surface glycoprotein that is characterized by its variants displaying differential overexpression in human cancers. Aptamers, also known as chemical antibodies, can target cell-surface molecules with high affinity and specificity via structural recognition. Aptamer-mediated drug delivery hence is of high potentials in guiding therapy to improve efficacy.
Methods: Variants CD44E and CD44s were studied for HCC relevance by investigating their expressions in primary HCC tumors, adjacent cirrhotic/fibrotic livers and normal livers using junction specific primers in qPCR assay. CD44E/s dual-targeted aptamers were uncovered by integrating loss-gain cell-SELEX and next generation sequencing. Selected aptamers were characterized for binding affinity and specificity, biostability, in vivo and in vitro cytotoxicity, in vivo homing and biodistribution, and ability to deliver 5-FU into targeted cells in vitro.
Results: Both CD44E and CD44s isoforms showed significant upregulations in HCC tumors with CD44E/s activities promoting cell proliferation and migration. Loss-gain cell-SELEX uncover a CD44E/s dual-targeting aptamer, termed CD44-Apt1. Strong binding of CD44-Apt1 to cell-surface CD44 positive cells but not CD44-negative cells was demonstrated by flow-cytometry. CD44-Apt1 displayed strong affinity to CD44E and CD44s with KD as low as 1 nM but not the hyaluronic acid binding domain of CD44. Confocal imaging of CD44-positive cells stained with fluorescent-labeled CD44-Apt1 showed profound cytoplasmic localization, suggesting efficient cell-penetrating ability. Meanwhile, no apparent staining was observed in CD44-negative cells. CD44-Apt1 when conjugated with inhibitor 5-FU showed efficient guidance of 5-FU into HCC cells that significantly enhanced drug toxicity by more than thousands-fold. Both in vitro cell treatment and in vivo animal biodistribution indicated that CD44-Apt1 is non-toxic. In HCC xenograft model, CD44-Apt1 efficiently homed to tumor xenografts in a CD44 expression-dependent manner.
Conclusion: Novel discovery of aptamer CD44-Apt1 that can bind both CD44E and CD44s illustrates high potential as nanoprobe to deliver anti-cancer therapeutics.
Keywords: CD44, aptamer, nanoprobe, hepatocellular carcinoma, targeted cancer therapy

 Global reach, higher impact
Global reach, higher impact