ISSN: 2206-7418
Nanotheranostics 2021; 5(4):488-498. doi:10.7150/ntno.62213 This issue Cite
Research Paper
Novel Protocols for Scalable Production of High Quality Purified Small Extracellular Vesicles from Bovine Milk
1. Fralin Biomedical Research Institute at Virginia Tech Carillion, Roanoke, VA, 24016, USA.
2. Center for Vascular and Heart Research, Virginia Tech, Roanoke, VA, 24016, USA.
3. Department of Biomedical Engineering and Mechanics, Virginia Tech, Blacksburg, VA, 24061, USA.
4. Department of Emergency Medicine, Virginia Tech Carilion School of Medicine, Virginia Tech, Roanoke, VA, 24016, USA.
5. Faculty of Health Science, Virginia Tech, Blacksburg, VA, 24061, USA.
* These authors contributed equally to this work.
Abstract
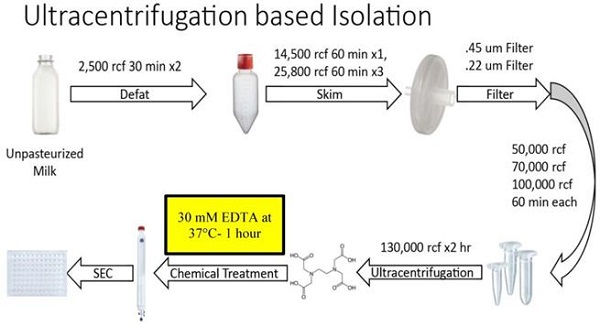
Extracellular Vesicles (EVs) are cell-secreted nanovesicles that have unique potential for encapsulating and targeting “difficult-to-drug” therapeutic cargos. Milk provides an enriched source of EVs, and of particular interest to the drug delivery field, small EVs. Small EVs are distinguished from large EVs by membrane components, biogenesis mechanism and downstream functionality - in particular, small EVs are primarily composed of exosomes, which show high stability in vivo and naturally function in the targeted delivery of biological materials to cells. Moreover, bovine milk is abundantly produced by the dairy industry, widely consumed, and generally well tolerated by humans. Importantly, there is evidence that milk exosomes and small EVs are efficiently taken up into the circulation from the gut, providing the opportunity for their use in administration of therapeutics such as microRNAs or peptides not typically available via an oral route. Unfortunately, present methods for isolation do not efficiently separate EVs from milk proteins, resulting in contamination that is not desirable in a clinical-grade therapeutic. Herein, we present novel EV purification methods focused on optimized timing and levels of temperature and divalent cation chelation. Incorporation of these solubilization steps into centrifugation- and tangential flow filtration-based methods provide large amounts of purified small EVs at ultra-dense concentrations, which are substantially free from contaminating milk proteins. Remarkably, these ultra-dense isolates equal 10 to 15% of the starting volume of milk indicating a prodigious rate of small EV production by mammary glands. Our approach enables gentle, scalable production of ultrastructurally and functionally intact small EVs from milk, providing a path to their industrial scale purification for oral delivery of therapeutic biologics and small drugs.
Keywords: Exosomes, Small Extracellular Vesicles, Bovine Milk Extracellular Vesicles, Small Extracellular Vesicle Isolation, Tangential Flow Filtration, Drug Delivery vehicle

 Global reach, higher impact
Global reach, higher impact