Nanotheranostics 2021; 5(2):125-142. doi:10.7150/ntno.52168 This issue Cite
Research Paper
MR-labelled liposomes and focused ultrasound for spatiotemporally controlled drug release in triple negative breast cancers in mice
1. School of Cancer & Pharmaceutical Sciences, King's College London, U.K.
2. Department of Academic Surgery, Imperial College London, U.K.
3. Department of Chemical Engineering, Imperial College London, U.K.
4. Department of Neuroimaging, King's College London, U.K.
#Contributed equally to this work.
Abstract
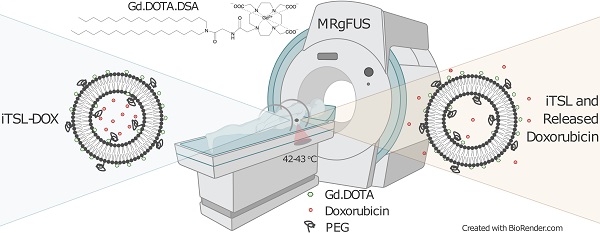
Rationale: Image-guided, triggerable, drug delivery systems allow for precisely placed and highly localised anti-cancer treatment. They contain labels for spatial mapping and tissue uptake tracking, providing key location and timing information for the application of an external stimulus to trigger drug release. High Intensity Focused Ultrasound (HIFU or FUS) is a non-invasive approach for treating small tissue volumes and is particularly effective at inducing drug release from thermosensitive nanocarriers. Here, we present a novel MR-imageable thermosensitive liposome (iTSL) for drug delivery to triple-negative breast cancers (TNBC).
Methods: A macrocyclic gadolinium-based Magnetic Resonance Imaging (MRI) contrast agent was covalently linked to a lipid. This was incorporated at 30 mol% into the lipid bilayer of a thermosensitive liposome that was also encapsulating doxorubicin. The resulting iTSL-DOX formulation was assessed for physical and chemical properties, storage stability, leakage of gadolinium or doxorubicin, and thermal- or FUS-induced drug release. Its effect on MRI relaxation time was tested in phantoms. Mice with tumours were used for studies to assess both tumour distribution and contrast enhancement over time. A lipid-conjugated near-infrared fluorescence (NIRF) probe was also included in the liposome to facilitate the real time monitoring of iTSL distribution and drug release in tumours by NIRF bioimaging. TNBC (MDA-MB-231) tumour-bearing mice were then used to demonstrate the efficacy at retarding tumour growth and increasing survival.
Results: iTSL-DOX provided rapid FUS-induced drug release that was dependent on the acoustic power applied. It was otherwise found to be stable, with minimum leakage of drug and gadolinium into buffers or under challenging conditions. In contrast to the usually suggested longer FUS treatment we identified that brief (~3 min) FUS significantly enhanced iTSL-DOX uptake to a targeted tumour and triggered near-total release of encapsulated doxorubicin, causing significant growth inhibition in the TNBC mouse model. A distinct reduction in the tumours' average T1 relaxation times was attributed to the iTSL accumulation.
Conclusions: We demonstrate that tracking iTSL in tumours using MRI assists the application of FUS for precise drug release and therapy.
Keywords: liposome, MRI, focused ultrasound, doxorubicin, triple-negative breast cancer

 Global reach, higher impact
Global reach, higher impact