Nanotheranostics 2021; 5(1):8-26. doi:10.7150/ntno.50633 This issue Cite
Research Paper
Neural precursors cells expanded in a 3D micro-engineered niche present enhanced therapeutic efficacy in vivo
1. Pediatric Clinical Research Center “Romeo and Enrica Invernizzi”, L. Sacco Department of Biomedical and Clinical Sciences, University of Milano, Milano, 20157, Italy.
2. Department of Chemistry, Materials and Chemical Engineering “Giulio Natta”, Politecnico di Milano, Milano, 20133, Italy.
3. Istituto di Fotonica e Nanotecnologie (IFN)-CNR and Department of Physics, Politecnico di Milano, Milano, 20133, Italy.
4. Genomic and Postgenomic Lab, IRCCS Mondino Foundation, Pavia, 27100, Italy.
Abstract
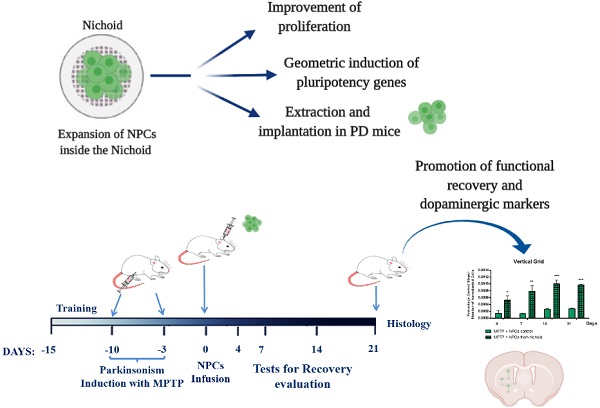
Rationale: Stem Cells (SCs) show a great potential in therapeutics for restoring and regenerating native tissues. The clinical translation of SCs therapies is currently hindered by the inability to expand SCs in vitro in large therapeutic dosages, while maintaining their safety and potency. The use of biomaterials allows for the generation of active biophysical signals for directing SCs fate through 3D micro-scaffolds, such as the one named “Nichoid”, fabricated with two-photon laser polymerization with a spatial resolution of 100 nm. The aims of this study were: i) to investigate the proliferation, differentiation and stemness properties of neural precursor cells (NPCs) following their cultivation inside the Nichoid micro-scaffold; ii) to assess the therapeutic effect and safety in vivo of NPCs cultivated in the Nichoid in a preclinical experimental model of Parkinson's Disease (PD).
Methods: Nichoids were fabricated by two photon laser polymerization onto circular glass coverslips using a home-made SZ2080 photoresist. NPCs were grown inside the Nichoid for 7 days, counted and characterized with RNA-Seq, Real Time PCR analysis, immunofluorescence and Western Blot. Then, NPCs were transplanted in a murine experimental model of PD, in which parkinsonism was induced by the intraperitoneal administration of the neurotoxin MPTP in C57/bl mice. The efficacy of engrafted Nichoid-expanded NPCs was evaluated by means of specific behavioral tests and, after animal sacrifice, with immunohistochemical studies in brain slices.
Results: NPCs grown inside the Nichoid show a significantly higher cell viability and proliferation than in standard culture conditions in suspension. Furthermore, we report the mechanical conditioning of NPCs in 3D micro-scaffolds, showing a significant increase in the expression of pluripotency genes. We also report that such mechanical reprogramming of NPCs produces an enhanced therapeutic effect in the in vivo model of PD. Recovery of PD symptoms was significantly increased when animals were treated with Nichoid-grown NPCs, and this is accompanied by the recovery of dopaminergic markers expression in the striatum of PD affected mice.
Conclusion: SCs demonstrated an increase in pluripotency potential when expanded inside the Nichoid, without the need of any genetic modification of cells, showing great promise for large-scale production of safe and functional cell therapies to be used in multiple clinical applications.
Keywords: two-photon laser polymerization, pluripotency, nichoid, 3D micro-scaffolds, regenerative medicine

 Global reach, higher impact
Global reach, higher impact