ISSN: 2206-7418
Nanotheranostics 2020; 4(4):210-223. doi:10.7150/ntno.49898 This issue Cite
Research Paper
Histologic evaluation of activation of acute inflammatory response in a mouse model following ultrasound-mediated blood-brain barrier using different acoustic pressures and microbubble doses
1. Department of Radiology, Stanford University, Stanford, California 94305, USA.
2. Department of Electrical Engineering, Stanford University, Stanford, California 94305, USA.
3. Department of Neurology and Neurological Sciences, Stanford University, Stanford, California 94305, USA.
4. Department of Comparative Medicine, Stanford University, Stanford, California 94305, USA.
*These authors contributed equally to this work.
Abstract
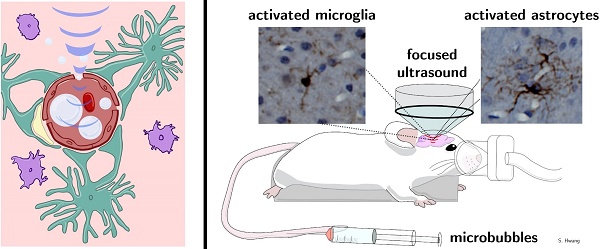
Rationale: Localized blood-brain barrier (BBB) opening can be achieved with minimal to no tissue damage by applying pulsed focused ultrasound alongside a low microbubble (MB) dose. However, relatively little is known regarding how varying treatment parameters affect the degree of neuroinflammation following BBB opening. The goal of this study was to evaluate the activation of an inflammatory response following BBB opening as a function of applied acoustic pressure using two different microbubble doses.
Methods: Mice were treated with 650 kHz ultrasound using varying acoustic peak negative pressures (PNPs) using two different MB doses, and activation of an inflammatory response, in terms of microglial and astrocyte activation, was assessed one hour following BBB opening using immunohistochemical staining. Harmonic and subharmonic acoustic emissions (AEs) were monitored for all treatments with a passive cavitation detector, and contrast-enhanced magnetic resonance imaging (CE-MRI) was performed following BBB opening to quantify the degree of opening. Hematoxylin and eosin-stained slides were assessed for the presence of microhemorrhage and edema.
Results: For each MB dose, BBB opening was achieved with minimal activation of microglia and astrocytes using a PNP of 0.15 MPa. Higher PNPs were associated with increased activation, with greater increases associated with the use of the higher MB dose. Additionally, glial activation was still observed in the absence of histopathological findings. We found that CE-MRI was most strongly correlated with the degree of activation. While acoustic emissions were not predictive of microglial or astrocyte activation, subharmonic AEs were strongly associated with marked and severe histopathological findings.
Conclusions: Our study demonstrated that there were mild histologic changes and activation of the acute inflammatory response using PNPs ranging from 0.15 MPa to 0.20 MPa, independent of MB dose. However, when higher PNPs of 0.25 MPa or above were applied, the same applied PNP resulted in more severe and widespread histological findings and activation of the acute inflammatory response when using the higher MB dose. The potential activation of the inflammatory response following ultrasound-mediated BBB opening should be considered when treating patients to maximize therapeutic benefit.
Keywords: blood-brain barrier, focused ultrasound, microbubbles, acute neuroinflammation, immunohistochemistry

 Global reach, higher impact
Global reach, higher impact