Nanotheranostics 2020; 4(2):83-90. doi:10.7150/ntno.43808 This issue Cite
Research Paper
Phospholipid Oxygen Microbubbles for Image-Guided Therapy
1. Department of Mechanical Engineering, University of Colorado, Boulder, CO, USA.
2. Department of Bioengineering, University of Colorado Anschutz Medical Campus, Aurora, CO, USA.
3. Department of Biomedical Engineering, University of North Carolina at Chapel Hill and NC State, Chapel Hill, NC, USA.
4. Biomedical Engineering Program, University of Colorado, Boulder, CO, USA.
Abstract
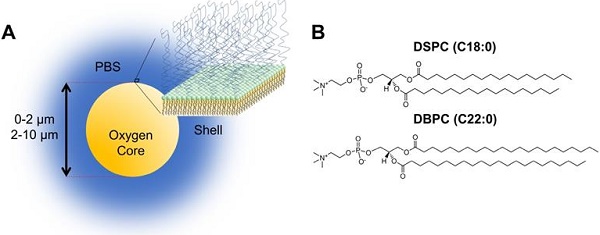
In recent work, oxygen microbubbles (OMB) have been shown to oxygenate hypoxic tumors, increase radio-sensitivity and improve tumor control by radiation therapy. Compared to intra-tumoral injection, intravenous delivery of adjuvant agents such as OMBs for radiotherapy offers an attractive means of achieving true theranostic function in a minimally invasive manner via contrast-enhanced ultrasound (CEUS), while reducing the risk of injury, infection or displacing tumor cells. However, short intravascular circulation times with conventional DSPC-lipid OMBs may lead to premature off-target dissolution of OMBs with an associated reduction in tumoral oxygen delivery. Prior work on microbubble stability and gas exchange suggests that increasing phospholipid acyl-chain length of the encapsulating shell and OMB size may increase circulation persistence, delivery and dissolved oxygen content. In the following studies, we investigate the effect of two phospholipid shell compositions, DSPC (C18:0) and DBPC (C22:0), as well as three size distributions (0.5-2 µm, 2-10 µm and polydisperse) on OMB circulation persistence utilizing CEUS in the kidneys of live C57B1/6 male and female mice, six weeks of age. DBPC OMB formulations demonstrated increased circulation half-lives versus DSPC formulations (2.4 ± 1.0 vs. 0.6 ± 0.5 s, p<0.01 for 2-10 µm), as well as an increased maximum intensity by over tenfold (p<0.01). Size-dependent effects remained consistent across both formulations with larger 2-10 µm microbubbles demonstrating significantly increased half-lives (2.4 ± 1.0 vs. 0.3 ± 0.2 s, p < 0.01) compared to smaller 0.5-2 µm formulations of DBPC. These studies indicate that DBPC 2-10 µm OMBs may be improved adjuvant agents for radiotherapy with significant potential for CEUS interrogation.
Keywords: DBPC, size isolation, ultrasound contrast agent, tumor hypoxia, radiation therapy

 Global reach, higher impact
Global reach, higher impact