Nanotheranostics 2019; 3(2):223-235. doi:10.7150/ntno.35935 This issue Cite
Research Paper
Overcoming hypoxia-induced chemoresistance to cisplatin through tumor oxygenation monitored by optical imaging
1. Department of Pharmaceutical Sciences, University of Connecticut, Storrs, Connecticut, USA 06269
2. Section of Cardiovascular Medicine, Department of Medicine, Yale University School of Medicine, New Haven, CT, USA 06519
3. Department of Cell Biology, University of Connecticut Health Center, Farmington, Connecticut, USA 06030
4. Department of Radiology, School of Medicine, University of North Carolina at Chapel Hill, North Carolina, USA 27599
* Equal contributors to this work.
#Current Address: Harvard T.H. Chan School of Public Health, Boston, Massachusetts, USA 02115
$ Current Address: Eli Lilly and Company Global Headquarters Lilly Corporate Center, Indianapolis, Indiana, USA 46285
Abstract
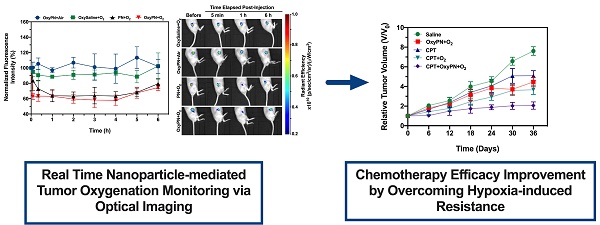
Perfluorocarbon nanoparticles have been reported to deliver oxygen to tumors and reduce hypoxia-induced radioresistance, however few studies have been carried out to study its role in reducing hypoxia-induced chemoresistance. The oxygenation effect also varies dramatically between different perfluorocarbon formulations and protocols, and there have been no efficient tools to monitor dynamic changes of tumor oxygenation non-invasively. Our goal was to promote tumor oxygenation using perfluorooctyl bromide (PFOB) nanoemulsion and to assess its role in sensitizing tumors to cisplatin treatment. A novel optical imaging protocol was also created to monitor the dynamic changes of tumor oxygenation in real-time.
Methods: PFOB nanoemulsion with high oxygen-carrying capacity was prepared and administered to tumor-bearing mice intravenously. Tumor oxygenation was monitored using optical imaging with a hypoxia probe injected intratumorally, thus the oxygenation dynamics and best oxygenation protocol were determined. Various treatment groups were studied, and the tumor growth was monitored to evaluate the role of oxygenation in sensitizing tumors to cisplatin treatment.
Results: PFOB nanoemulsion with and without pre-oxygenation along with carbogen breathing resulted in much better tumor oxygenation compared to carbogen breathing alone, while PFOB with air breathing did not show significant increase in tumor oxygenation. Pre-oxygenated PFOB with carbogen breathing produced the most effective oxygenation as early as 5 min post administration. In vitro and in vivo data showed preoxygenated PFOB nanoemulsion with carbogen breathing could increase cisplatin-mediated apoptosis of cancer cells and inhibited tumor growth at a low dose of cisplatin (1 mg/kg) treatment. Furthermore, the treatment did not induce nephrotoxicity.
Conclusions: Preoxygenated PFOB nanoemulsion with carbogen breathing can effectively increase tumor oxygenation, which has a great potential to prevent/overcome hypoxia-induced chemotherapy resistance. In addition, optical imaging with intratumoral injection of the hypoxia probe was an efficient tool to monitor tumor oxygenation dynamics during PFOB administration, providing better understanding on oxygenation effects under different protocols.
Keywords: perfluorooctyl bromide, hypoxia, oxygen delivery, cisplatin, chemoresistance.

 Global reach, higher impact
Global reach, higher impact