ISSN: 2206-7418
Nanotheranostics 2018; 2(3):280-294. doi:10.7150/ntno.25555 This issue Cite
Research Paper
HAase-sensitive dual-targeting irinotecan liposomes enhance the therapeutic efficacy of lung cancer in animals
Institute of Environment and Sustainable Development in Agriculture, Chinese Academy of Agricultural Sciences, Beijing, China
Nanobiotechnology Research Center, Chinese Academy of Agricultural Sciences, Beijing, China
Abstract
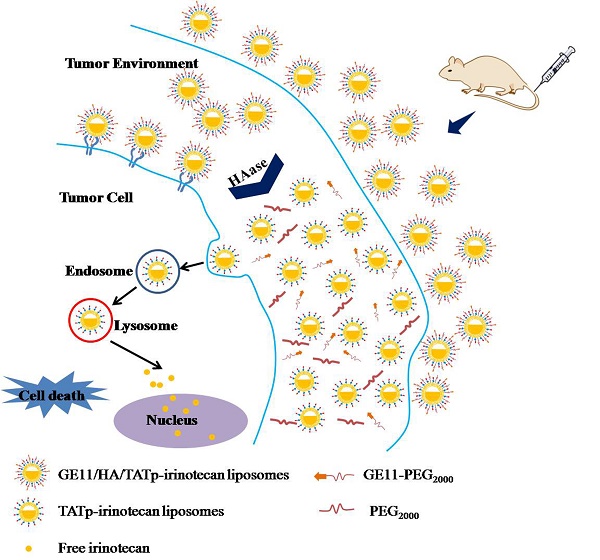
Among all cancers, lung cancer is one of the most common and serious types of cancer. It is challenging for site-specific delivery of anticancer therapeutics to tumor cells. Herein, we developed a novel“smart” dual-targeting liposomal platform to respond to the highly expressed hyaluronidase (HAase) in the tumor microenvironment and improve tumor targeting and antitumor efficacy.
Methods: In our design, the HA was used as a sensitive linker between a liposomal lipid and long chain PEG block to synthesize three functional conjugates in order to prepare“smart” liposomal platform modified with epidermal growth factor receptor (EGFR) antibody (GE11) and cell-penetrating peptide (TATp). Using irinotecan as a model therapeutic, evaluations were performed on the human lung adenocarcinoma A549 cells as well as the xenografted A549 cancer cells in nude mice.
Results: The GE11/HA/TATp-irinotecan liposomes evidently increased the uptake of irinotecan and showed significant antitumor efficacy in the xenografted A549 cancer cells in nude mice by intravenous administration. The mechanisms were defined to be two aspects: GE11 exhibits high affinity for EGFR binding and the degradation of the HA by HAase results in the long-chain PEG removal and exposure of the previously hidden surface-attached TATp to enhance the target cell internalization.
Conclusion: Our findings suggest that this functional liposomal platform may provide a novel strategy for treating lung cancers because of effective intracellular delivery.
Keywords: Lung cancer, Hyaluronic acid (HA), HAase-sensitive dual-targeting irinotecan liposomes, Antitumor efficiency, Intracellular delivery

 Global reach, higher impact
Global reach, higher impact